In silico analysis is a natural complement to in vivo and in vitro testing. It uses engineering simulation to create reliable, computer-based models that predict interactions among medical devices, drugs and the human body. The first models, created in the late nineties, were rudimentary but valuable. Since 2000, however, engineering simulations have made tremendous progress in both computation time required and fidelity of models to the complex reality of human tissues.
You might also be interested in ‘Simulate your trials and save time and money’

Figure: Treating patient-specific aneurysm by bringing simulations into the operating room (Courtesy of Sim-and-Cure and Prof V. Costalat, Neuroradiology Department, Montpellier Hospital)
Slowly but surely
Despite obvious advantages of in silico analysis, general acceptance of this technique by legislative authorities proceeded slowly. This is due to three major challenges:
- Opening the minds of medical device, biotech, and pharmaceutical companies.
The healthcare world is very tentative about in silico testing. However, the need for medical innovations that treat patients better and faster, the tightening of regulations to maximize patient safety, and pressure from both local governments and patients to reduce the skyrocketing cost of healthcare, were impossible to address without a technological shift. The industrial world evaluated and quickly embraced engineering simulation technology. - Developing reliable models.
Twenty years ago, the first models produced good qualitative assessments of what is likely to happen in the human body. However, when peoples’ lives are at stake, this is not good enough. The last two decades have witnessed an unprecedented academic and industrial collaboration to evolve and continuously evaluate new models that can be personalized to any specific patient. The recent adoption of simulations by a growing number of clinicians in the operating room confirms this trend. - Gaining support from regulatory authorities.
If agencies such as the Food and Drug Administration (FDA), European Medicines Agency (EMA), China Food and Drug Administration (CFDA), and Product Development and Management Association (PDMA) are willing to accelerate medical innovation, they need to solidly guarantee patients’ safety. These regulatory authorities carefully investigated, challenged, assessed, and progressively accepted engineering simulation as an essential component of any product submission. Many agencies, inspired by the early work and communication of the FDA, are now encouraging the strictly controlled use of computer models throughout their approval processes.
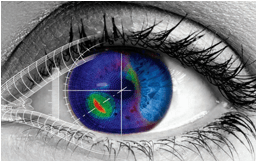
Figure: An advanced eye model to predict the results of laser therapy (Courtesy of Optimo Medical)
The European Parliament voted Yes!
More and more healthcare professionals, academics, industry leaders, regulatory authorities, clinicians, and even patients, are progressively recognizing the crucial role played by in silico analyses in the healthcare product development process. But for the in silico approach to be legitimized, legislative authorities need to carefully assess it and take the necessary actions to formally regulate it and encourage its controlled use. This is one of the ambitious goals of the Avicenna Alliance.
The Avicenna Alliance has been working with supportive members of the European Parliament to modernize elements of the 2004 EMA Regulation. This will encourage the in silico market and provide certainty for investors and researchers in the in silico community. On March 10, 2016, the European Parliament voted — with an overwhelming majority — in favor of requiring the EMA to take into account alternative models in clinical trials, such as ‘human-relevant computer or cellular models, pathways of toxicity, or adverse outcome pathways.’
Although this is a great first step, there still is a long way to go. To accelerate the wide adoption of in silico analyses, help is needed from all healthcare professionals. You can contact the Avicenna Alliance directly to find out the best way to contribute.
Drafted by Thierry Marchal, Ansys
You might also be interested in ‘Simulate your trials and save time and money‘


